Centre for Drugs and Diagnostics
Innovating drugs and diagnostics to defeat infectious diseases

The Centre for Drugs and Diagnostics (CDD) at LSTM comprises an experienced multi-disciplinary team of experts working to discover, develop, and deliver novel therapies and diagnostics against a range of pathogens.
Infectious diseases continue to pose significant threats to global health, particularly affecting vulnerable populations in low-resource settings. Many of these diseases lack adequate diagnostic tools and effective treatments, while emerging pathogens and antimicrobial resistance (AMR) create new challenges that require innovative solutions.
Building on more than 25 years of translational research, the CDD works with industry, academia, and nongovernment organisations to accelerate scientific discovery into real-world application and impact. In the past four years CDD has been involved in bringing 18 products to market to diagnose and treat a wide range of diseases.
Our work spans the complete translational pipeline, from finding new drug targets and testing promising compounds to running clinical trials and ensuring that innovations reach communities.
Our research focus and approaches
We discover and develop new medicines for diseases that affect the world’s underserved communities
Our teams work in state-of-the-art laboratories to find and create new drugs for diseases such as malaria, tuberculosis, and neglected tropical diseases (NTDs). Working closely with partners at the University of Liverpool, we take promising compounds from early discovery all the way through to clinical trials. Our focus is on diseases that disproportionately affect people in low-resource settings, where safe and effective treatments are urgently needed.
We create rapid tests that can diagnose multiple diseases at once
We develop innovative diagnostic tools that can detect several diseases from a single test, even in places without traditional laboratory facilities. These rapid tests enable healthcare workers to quickly identify the cause of an individual’s illness and start the appropriate treatment faster. Our portable diagnostic technologies are designed to work in remote areas where access to medical testing is limited.
We ensure our innovations reach the people who need them most
Beyond discovering new drugs and tests, we work with governments, health authorities, and companies to make sure our breakthroughs reach patients. We focus on creating solutions that are affordable, easy to use, and practical for health systems with limited resources. Our goal is to turn scientific discoveries into real improvements in people’s health and lives.
Our team

Professor Joseph Turner
Joseph Turner researches the immunobiology and chemotherapy of helminth neglected tropical diseases. A major goal of his research is the clinical translation of new therapeutics and diagnostics for filarial neglected tropical diseases which are prioritised for global elimination.

Professor Giancarlo Biagini
A recognised international leader in translational infection research, Giancarlo has led major global health consortia and contributes to shaping national and international research agendas.

Dr Ana Cubas Atienzar
Dr Atienzar is a Lecturer at LSTM specializing in developing and evaluating rapid point-of-care diagnostics for emerging infectious diseases, including viral haemorrhagic fevers and COVID-19.
Our impact
We have developed breakthrough diagnostic technologies
LSTM has developed the world’s first rapid test for Crimean-Congo haemorrhagic fever (CCHF), a deadly tick-borne disease with mortality rates of up to 40 percent. The lateral flow test delivers results in just 30 minutes without laboratory equipment, replacing PCR tests that can take up to 10 days, enabling faster diagnosis, earlier treatment, and better outbreak control. Alongside this, LSTM is pioneering tests that can simultaneously detect multiple arbovirus infections, including dengue, Zika, chikungunya, and Mayaro, helping health workers quickly identify the cause of fever and begin treatment.
We are advancing novel therapeutic approaches across multiple disease areas
Our drug discovery programmes have contributed to significant advances in treating malaria, tuberculosis, NTDs, and antimicrobial-resistant infections. A∙WOL antifilarial drug discovery team won the prestigious Royal Society of Chemistry Prize in 2024, demonstrating the international recognition of our innovative approaches to drug development.
We strengthen capacity and partnerships for sustained impact
A recent £10 million award from UKRI’s Expanding Excellence in England (E3) fund to expand our centre reflects our growing capacity to train the next generation of researchers and strengthen our collaborative networks worldwide. We work with partners across sub-Saharan Africa, South Asia, and beyond to build local expertise and ensure that innovations we develop together are sustainably implemented where they can have the greatest health impact.
CDD Facilities
CDD researchers have access to world-leading facilities to accelerate translational research. This includes a 12-bed inpatient human challenge facility, providing a rapid and cost-effective platform for assessing vaccines and therapeutics.
Our capabilities are expanding through major investment. Supported by UKRI’s E3 fund and the Liverpool City Region Life Sciences Innovation Zone, we are developing a £12.5 million AI-enabled, robotic, high-containment laboratory to accelerate the pathway from discovery to deployable products.
Alongside this, the Biologics Regional Innovation Technology Ecosystem (BRITE) connects universities, industry and regional partners to translate research into new healthcare technologies, driving economic growth and delivering tangible health benefits.

Projects

MMVSola
A free tool that predicts how new malaria drugs will work in patients
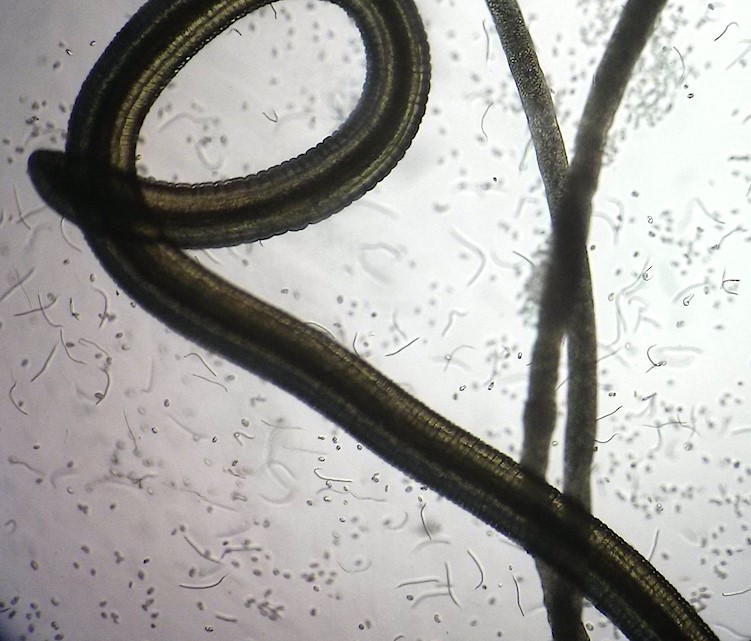
FLiP (Filariasis: Lipids in Pathogenesis)
FLiP is a five-year research programme which will will explore the therapeutic potential of targeting bioactive lipids to create new treatments for filariasis

Malaria in Pregnancy Consortium (MiP)
The Malaria in Pregnancy (MiP) Consortium is the world’s largest international research collaboration dedicated to protecting mothers and babies from malaria.